cgmaxed
Well-Known Member
I wondered the same thing. How was the fire not detected?What no smoke and fire alarms allowed in a Fire Station?
I wondered the same thing. How was the fire not detected?What no smoke and fire alarms allowed in a Fire Station?
I didn't look at the article. I didn't realize it started in a vehicle. My comment can be disregarded.@maelstrom - I expect they may cause an increase in battery temperature due to the containment but the Bat-Safe has metal walls that I imagine would conduct some of that away, and as long as the battery is reasonably healthy would withstand the amount of heat generated without causing runaway. If a battery is that close to having thermal runaway it should be showing signs of its condition, which should be an indicator either not to charge it or to watch it closely.
@cgmaxed - From the photos, I'd say the charging was taking place inside the centre vehicle. I'm guessing that there was no smoke detector inside the vehicle and the way those things burn, a significant amount of damage may have already been done by the time it was detectable, especially if one faulty battery then set others alight.... I couldn't see any fire suppression systems in the garage itself in the photos of the wreckage.
I was charging a 6S 1500 Mah battery when this happened. Glad I was using the Bat Safe. A lipo bag (I have several) would not have contained this.Will charging bags prevent this? How about a Bat-Safe?
I was thinking, I bet a fire safe would be a good place to put a charging lipo. Even with the lid open, a hard firesafe would contain the fire pretty well, unless sparks and hot pieces started flying out of the safe. Keeping the lid shut would definitely keep the fire contained, until it burned itself out.I was charging a 6S 1500 Mah battery when this happened. Glad I was using the Bat Safe. A lipo bag (I have several) would not have contained this.
Not surprised... those batteries have no charge protection whatsoever.I was charging a 6S 1500 Mah battery when this happened. Glad I was using the Bat Safe. A lipo bag (I have several) would not have contained this.
Thanks eEridani,@br14nh
Nice build, but don't see anything for overpressure or toxic gas mitigation. Did I miss something?
 www.bat-safe.com
www.bat-safe.com
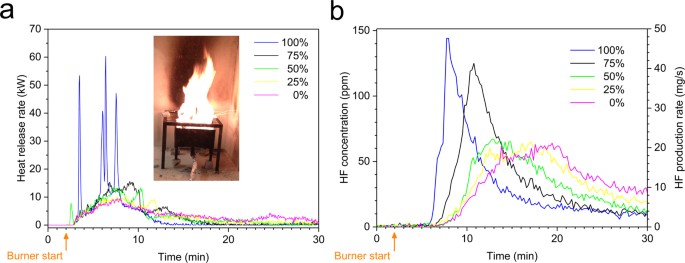
Our quantitative study of the emission gases from Li-ion battery fires covers a wide range of battery types. We found that commercial lithium-ion batteries can emit considerable amounts of HF during a fire and that the emission rates vary for different types of batteries and SOC levels.
The photo from earlier showed three fairly large batteries - 22.2v 1.5Ahr - 100Whr total - and with Lithium being the main ingredient I'm still concerned the HF byproduct concentration is dangerous.@eEridani - Thanks for those articles, very helpful.
I agree that wasn't a very bright thing to post on a company website [snip]
The thing that surprises me is how willing people are to handle burned battery residue, with no gloves, getting it on their clothing, etc. If I was dealing with a burnt battery I would want to be wearing disposable gloves, not touch anything else and wash my hands thoroughly after glove removal prior to touching my face, or doing anything else really.
How do these boxes and bags allow the heat generated during charging to be dissipated? Many lithium battery fires have been as a result of thermal runaway rather than internal shorting
While enclosing batteries in a container/box may also trap some heat, a healthy battery will stay well within a safe range. A box will allow a little more room for the heat to dissipate than a bag and the walls of commercial Bat Safe boxes are metal, which may help conduct some of that away.My thoughts as well, wouldn't the heat build-up from enclosing the charging batteries be more likely to cause an issue, rather than the recognised, but extremely unlikely, chance of battery faults causing a fire?
You can charge them outside in a large metal trunk that locks and is fixed to the slab so no one can steal them. Then they won’t burn down the house again.I used to be a manager in the semiconductor industry and one of the devices we made was widely used as a charge/discharge current limiter of lithium batteries. That kind of control helps against abuse of the battery but doesn't prevent an internal fault. Early lithium battery designs had plates that could warp and short out, causing catastrophic failure. Modern designs are better for that but not perfect. Most research I've seen into new battery technology involves not only higher energy density, but also materials that won't warp ... or plate separators that prevent shorts.
You can charge them outside in a large metal trunk that locks and is fixed to the slab so no one can steal them. Then they won’t burn down the house again.
I got this one from Home Depot for $150-$200.
Totally kills the problem and I can charge the whole fleet at once. No smoke in the house, garage or shop is possible.
But, as someone pointed out earlier, they have an internal seal. Any gases venting from the battery during charging will pressurise the box. If it does catch fire, the overpressure could be lethal.Or a cheap Ammo Box...... https://amzn.to/3Gtv8BE
Well I just learned a whole bunch. I was researching those lipo bags on Amazon and just came here for the truth. I'm going to retrofit a 50 cal metal ammo box tonight. So are the holes on top in case the battery goes up in flames or for heat dissipation while charging? I've been charging my batteries all over the place including my SUV when on a long shoot. Holy crap! I've been playing Russian battery roulette.I don't trust the bags as I don't believe any of them are robust enough.
I use a Bat-Safe box. When the last Covid lockdown prevented my buying another one (couldn't get them into Aus from anywhere at that stage) I bought some ammo cases and a steel toolbox and modified them to serve the same purpose - holes drilled in the lids with fibreglass matting filters and gyprock lining, they may not quite be up to Bat-Safe specs but way better than having nothing. I use them to charge all my LiPo batteries and store the RC ones as they will at least contain the heat from any fire enough to prevent it spreading. I also still monitor whenever charging.
We use essential cookies to make this site work, and optional cookies to enhance your experience.


